India’s Gennova Biopharmaceuticals is allegedly working to develop an “omicron-specific” Chinese coronavirus vaccine candidate based on mRNA technology that could be available to the public as soon as February, Reuters reported on Monday citing an unnamed company spokesman who confirmed the development via text message.
“The Omicron-specific variant of the vaccine is under development and will be ready for human clinical trials, subject to regulatory approvals,” a Gennova Biopharmaceuticals spokesman wrote in a text message to Reuters on January 17.
The news agency said it received the text from Gennova Biopharmaceuticals after “a person with direct knowledge of the matter said the product could be ready in a month or two.”
Gennova Biopharmaceuticals will launch human trials of its new mRNA vaccine candidate in February, according to a January 17 report by Asian News International (ANI).
“The country’s first messenger mRNA vaccine is expected to begin trials on humans in February,” ANI, India’s largest TV news agency, reported on Monday citing unnamed sources.

A health worker poses at a vaccination center administrating third ‘booster’ doses for the Covid-19 coronavirus in New Delhi on January 10, 2022, as the country sees an Omicron-driven surge in cases. ( SAJJAD HUSSAIN/AFP via Getty Images)
“The Pune-based Gennova Biopharmaceuticals has submitted phase 2 data of mRNA vaccine and has also completed the recruitment of phase 3 data. Drugs Controller General of India’s (DCGI) Subject Expert Committee (SEC) is expected to review the data soon,” the anonymous sources said.
The Gennova Biopharmaceuticals vaccine candidate in question would specifically target the omicron variant of SARS-CoV-2, which is a type of coronavirus that causes the disease known as “COVID-19,” or the Chinese coronavirus.
An mRNA vaccine is based on new technology and works differently than a traditional viral vector vaccine. The U.S. Centers for Disease Control and Prevention (CDC) explained how mRNA vaccines function in a statement posted by its website on January 4.
“To trigger an immune response, many vaccines put a weakened or inactivated germ into our bodies,” the CDC wrote. “Not mRNA vaccines. Instead, mRNA vaccines use mRNA created in a laboratory to teach our cells how to make a protein—or even just a piece of a protein—that triggers an immune response inside our bodies.”
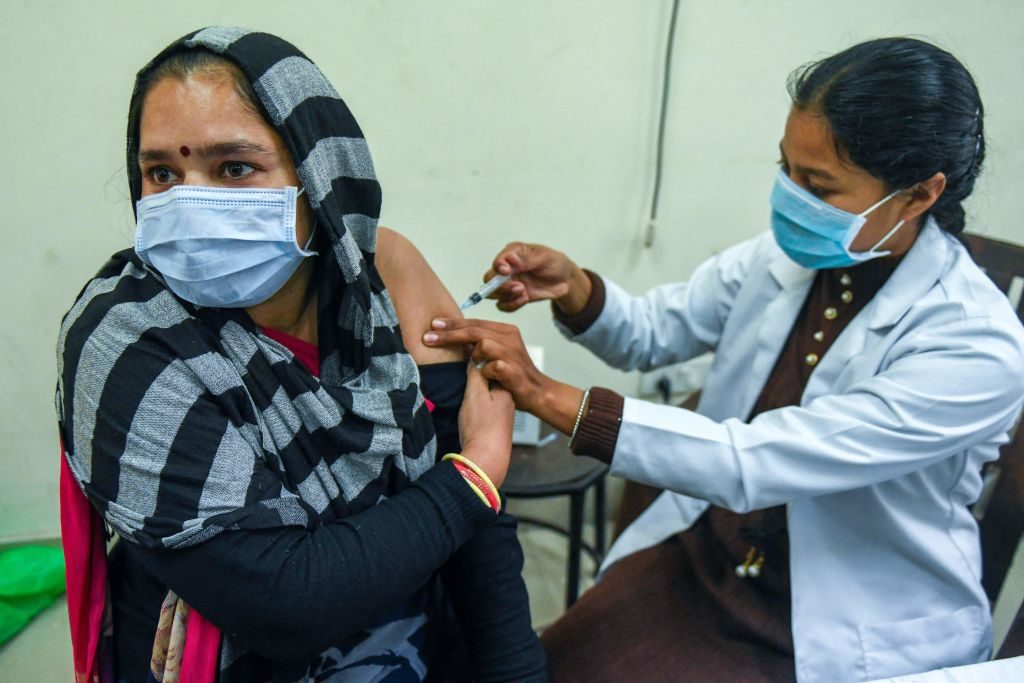
A health worker inoculates a woman with a third ‘booster’ dose of the Covid-19 coronavirus vaccine in a hospital in Amritsar on January 10, 2022, as the country sees an Omicron-driven surge in cases. (NARINDER NANU/AFP via Getty Images)
“That immune response, which produces antibodies, is what protects us from getting infected if the real virus enters our bodies,” according to the U.S. government health agency.
The U.S.-based pharmaceutical giant Pfizer currently offers an mRNA vaccine against the Chinese coronavirus that it developed together with German biotech company BioNTech and Chinese company Fosun Pharma. Moderna, another U.S.-based pharmaceutical company, has also produced an mRNA-based Chinese coronavirus vaccine. Neither Pfizer-BioNTech nor Moderna has yet developed an omicron-specific mRNA vaccine.
Widespread use of the companies’ current coronavirus vaccine offerings has done little to prevent the spread of omicron, which is more capable of evading immunity imparted by vaccines than previous strains of SARS-CoV-2.

COMMENTS
Please let us know if you're having issues with commenting.